
- This event has passed.
Dissertation Proposal - Kalin Gibbons
February 16, 2023 @ 8:30 am MST
 Development of an Inferential Statistics Model of the Natural Knee for Real-time Prediction of Joint Mechanics
Development of an Inferential Statistics Model of the Natural Knee for Real-time Prediction of Joint Mechanics
Presented by Kalin Gibbons – Data Science emphasis
In-person – MEC 201
Abstract
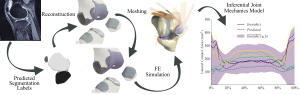
Knee injury and disease represent a significant global burden on the world’s medical system and result in reduced quality of life for millions of people worldwide. The knee joint is a primary component of the musculoskeletal system that absorbs and redirects large forces during activities of daily living and is often subjected to acute injury or degenerative disease. Knee osteoarthritis (OA) is the most common form of arthritis and the leading cause of ambulatory disability, which often necessitates the use of knee implants in later stages. OA is a multifactorial disease resulting in morphological and biochemical tissue adaptations recorded in the cartilage, ligaments, synovial fluid, and bone. Changes in joint biomechanics due to aging or traumatic injury are thought to be a major driver of these adaptations, but researchers are limited in their ability to collect in vivo mechanical data from the natural knee. Validated computational studies aim to fill this gap, but several preprocessing bottlenecks limit researchers to small cohorts of subjects or parameterized knee implant geometries. Working around these limitations, past researchers have been able to map geometric features to implant mechanics during a simulated knee bend activity using classical inferential statistics. Generating results using these statistical models requires orders of magnitude less time and expertise than developing a working finite element (FE) simulation, which can help encourage clinical adoption of computational model driven, individualized patient care. The main barriers preventing similar studies with the natural knee are medical image segmentation and automatic hexahedral meshing of joint tissues, which can take days of person-hours per knee. Recent advances in deep-learning segmentation and the availability of prototype knee joint meshing software have presented us with an opportunity to eliminate these bottlenecks. The objectives of this proposed research are 1) to integrate automatic segmentation, hexahedral meshing, and soft-tissue ligament attachment locator algorithms into a medical image sequence to FE simulation pipeline capable of generating 100s of simulation-ready meshes 2) to produce the first population-scale computational study capable of generating subject-specific biomechanical data for the natural knee and 3) to use the generated data to develop an inferential statistics model mapping subject knee anatomy to resulting joint biomechanics.
Committee:
Dr. Clare Fitzpatrick, Dr. Trevor Lujan, Dr. Edoardo Serra, Mark Taylor