Koby W. Kimball, Alex Soto, Lexi Bray, Dr. Eric Ortlund, Dr. Brad Morrison
Torin 1 Restores Autophagy Through Lipid-Screening Inhibition
Background:
Our lab is interested in Parkinson’s Disease. One hallmark of PD is the accumulation of toxic protein aggregates made up of alpha-synuclein known as Lewy Bodies. It is believed that these protein aggregates are cleared from the cell via autophagy, a cellular process in which proteins, organelles, other cellular debris are engulfed in a vesicle (autophagosome) and degraded following fusion with a lysosome (Morrison). Despite many advances being made in the study of autophagy and PD, The PD community has yet to find any disease modifying treatments. With this in mind, we have decided to shift our focus towards lipid signaling, a topic that has not yet been adequately addressed. We recently performed an unbiased lipidomic screen of factors that affect autophagy and identified Stearic Acid, Hydroxystearic acid, and 5-OXO-ETE as autophagy inhibitors. After validation of these lipids, we set out to test the ability of an autophagy inducing drug, Torin 1 to rescue the autophagy inhibition associated with treatment of these lipids.
Methods:
Cell culture:
- Treat SH-SY5Y cells with Retinoic acid for 7 days.
- Treat cells with 2 different doses of fresh Torin 1 (250nM or 1 uM) or HBSS (positive control) with and without Bafilomycin A1 in media prepared with freshly thawed 15% FBS for 4 hours.
- Lyse and sonicate cells and then perform western blot.
Western blot:
- Run samples on a 16% Tris glycine gel for 90 minutes at 130 volts.
- Transfer samples via iBlot2 machine for 20 minutes at 10 volts.
- Membrane was probed for LC3-II and B-actin.
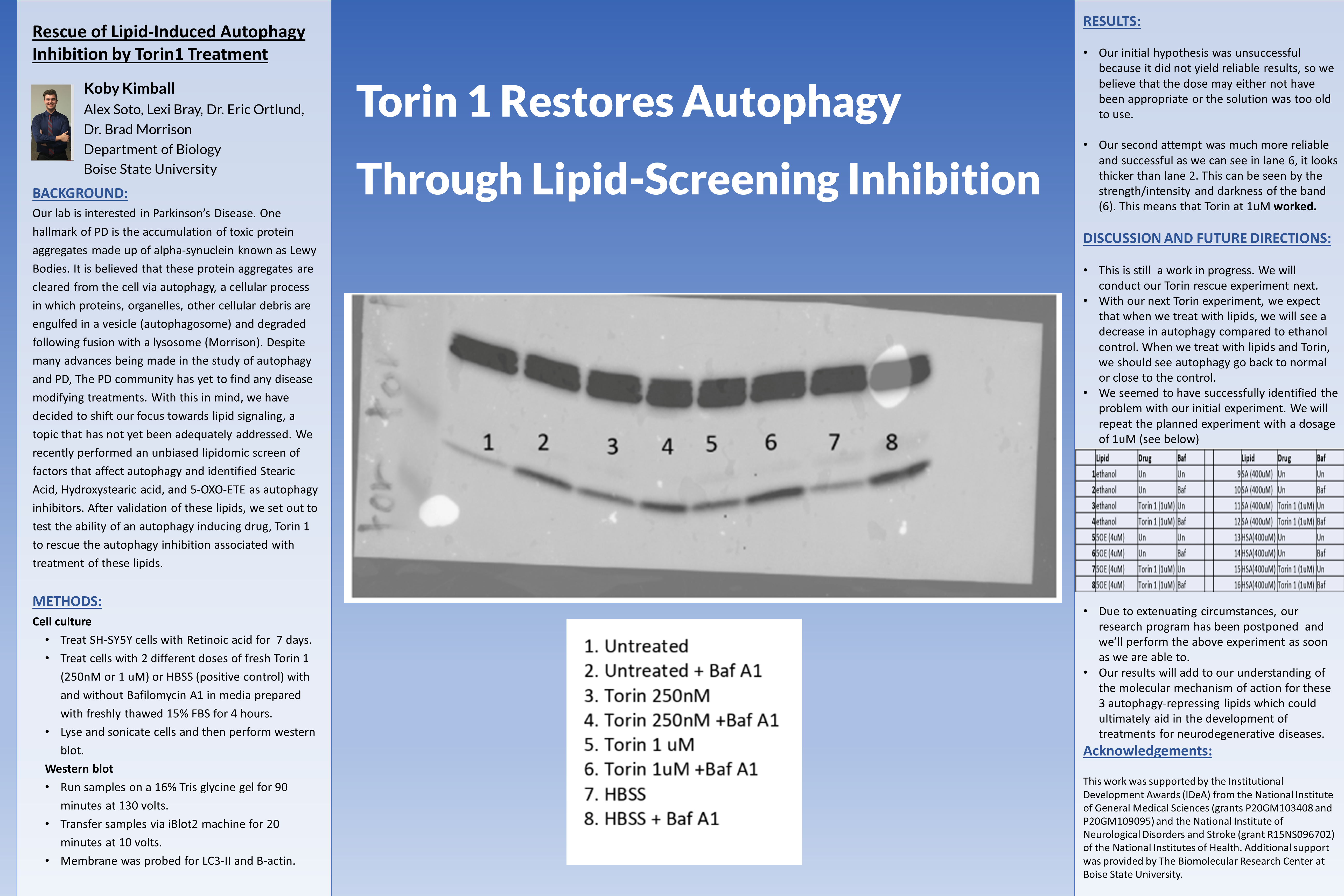
Results:
- Our initial hypothesis was unsuccessful because it did not yield reliable results, so we believe that the dose may either not have been appropriate or the solution was too old to use.
- Our second attempt was much more reliable and successful as we can see in lane 6, it looks thicker than lane 2. This can be seen by the strength/intensity and darkness of the band (6). This means that Torin at 1uM worked.
Discussion and Future Directions:
- This is still a work in progress. We will conduct our Torin rescue experiment next.
- With our next Torin experiment, we expect that when we treat with lipids, we will see a decrease in autophagy compared to ethanol control. When we treat with lipids and Torin, we should see autophagy go back to normal or close to the control.
- We seemed to have successfully identified the problem with our initial experiment. We will repeat the planned experiment with a dosage of 1uM (see below)
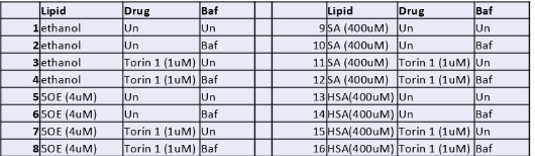
- Due to extenuating circumstances, our research program has been postponed and we’ll perform the above experiment as soon as we are able to.
- Our results will add to our understanding of the molecular mechanism of action for these 3 autophagy-repressing lipids which could ultimately aid in the development of treatments for neurodegenerative diseases.
Acknowledgements:
This work was supported by the Institutional Development Awards (IDeA) from the National Institute of General Medical Sciences (grants P20GM103408 and P20GM109095) and the National Institute of Neurological Disorders and Stroke (grant R15NS096702) of the National Institutes of Health. Additional support was provided by The Biomolecular Research Center at Boise State University.
Additional Information
For questions or comments about this research, contact Koby Kimball at kobykimball@u.boisestate.edu.